
This blog post answers the questions of what rare diseases are in general, in which medical areas they’re involved, how many people are affected and the number of medications, which are available now.
Definition and characteristics
This collective term stands for many different diseases which don’t share a consistent pathology.1,2 All these mulit-systemic diseases have in common that they’re chronic, progressive, degenerative and life-threatening.2 The quality of life is usually limited and there’s no effective cure.2
In Europe, a disease is defined as an rare disease if there aren’t more than 5 in 10,000 patients suffering from them.1 Due to the lack of information regarding the cause and course of the disease, 40% of all patients receive a misdiagnosis and thus no effective therapy.3 On average, it takes five years to get a correct diagnosis.3
Worldwide there’re about 8,000 rare diseases.3 However, due to the better understanding in human genetics, the number of known diseases is increasing annually.4,5
Cause
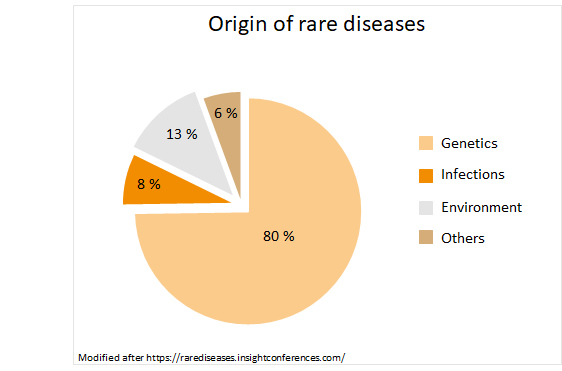
8 out of 10 diseases are triggered by a genetic defect.1,5,6 Due to the genetic anomalies a whole cascade of complaints are released, often leaving the source uncovered.3 However, meanwhile there’re clear assignments for 6,084 rare diseases, which are associated with around 4,000 genes (as of 2016).5 Rare diseases can also be caused by infections or environmental influences, often it’s also an interaction of several factors.6
Medical areas
Rare diseases are involved in many medical areas:
- Cancer: acute lymphocytic leukemia,4 renal cell cancer,7 hepatocellular carcinoma,7 carcinoma of the adrenal cortex7
- Cardiovascular diseases: pulmonary hypertension1
- Metabolic disorders: cystic fibrosis, phenylketonuria, Fabry disease4
- Neurological disorders: Neuronal ceroid lipofuscinosis,1 narcolepsy7
- Infectious diseases: tuberculosis,4
- Autoimmune diseases: lupus, Sjögren syndrome1
Selected samples for rare diseases
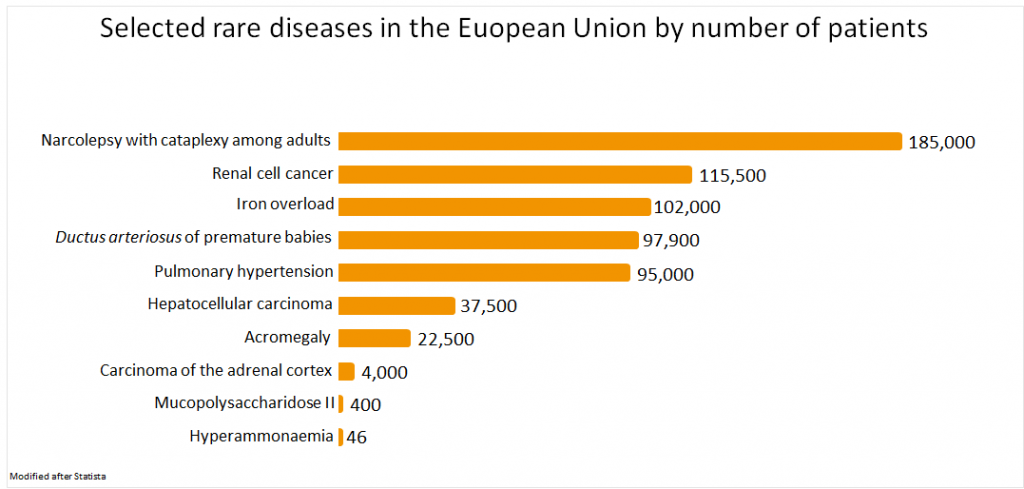
An example for a rare disease is narcolepsy with cataplexy among adults. About 185,00 people are affected in Europe.7 It’s a lifelong neurological disorder in which the sleep wake rhythm is disturbed.8 This is why it’s also called sleeping sickness.8 One of the symptoms is catalepsy.9 Patients suffer from short episodes of muscle failure, such as relaxation of the facial muscles or a complete collapse of the entire body.9 In contrast to epilepsy, those affected are fully conscious.9 The reason for this disease is a combination of genetic predispositions and environmental influences.10 Recent studies have revealed that there’s a lack of the neurotransmitter hypcretin in patients suffering from narcolepsy.10 This substance transmits signals from nerve cell to nerve cell and is involved in the regulation of the sleep-wake cycle.10 Misregulation of the immune response is thought to destroy the cells producing hypcretin.10 Narcolepsy occurs in 4 out of 10,000 people.11
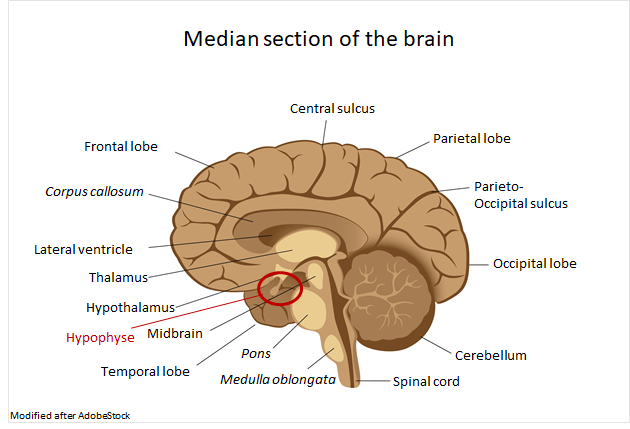
Another example is acromegaly,7 also called gigantism,12 which is responsible for changing the external appearance, metabolism and internal organs.13 The disease is caused by a tumor in the Hypophyse leading to an overproduction of growth hormones.12,13 Stimulation of IGF-1 in the liver promotes the growth of cartilage, bones, muscles and connective tissue.13 Among other things, this leads to an enlargement of hands, feet and tongue as well as facial features.13
Epidemiology
Worldwide there’re 410 million people affected by rare diseases.6 30 million are living in Europe,5,6 corresponding to 6-7% of the European population.4 At least half of all affected people are children.1,2,5 30% die before the age of five years.2
Comparision of rare and frequent diseases1

Orphan drugs
For a long time there was no research in the field of rare diseases, that’s why they are also called orphan diseases. The drugs for the treatment are referred as orphan drugs.1
Challenges in the development
In order to be able to develop effective drugs for certain diseases, it’s necessary to understand all related molecular processes leading to the disease.1 However, one characteristic of rare diseases is the lack of knowledge regarding the progression of the disease.1 Thus, basic research is needed in this field in order to be able to find and produce appropriate drugs.1 Furthermore, drug testing in clinical trials is difficult because often there aren’t enough patients for the study due to the rarity of the disease.1 Additionally this process is aggravated by the fact that many people don’t know about their disease because of the lack of knowledge of the physicians.1
Orphan drug status
In order to acquire an orphan drug status, the related disease have to fullfill following characteristics:1
- The disease have to be rare1
- The disease have to be severe, this means life-threating or leading to chronic disability.1
- The drug must have an additional benefit.1
The European Medicines Agency (EMA) decides if a drug gets the orphan drug status or not.1
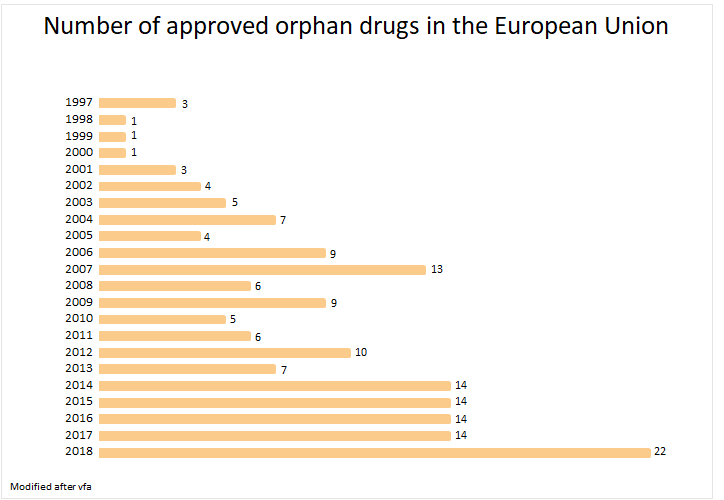
Orphan drug regulation
Developing medicines for rare diseases is an economical risk, because it involves time-consuming research, low chances of success and low turnover potential.1 A regulation introduced in the year 2000 in Europe and 1983 in the USA should improve that.1 The orphan drug regulation protects an approved drug for ten years against generic. Generic drugs which don’t differ in their composition and mode of action from the original drug are therefore not approved.1
Progress
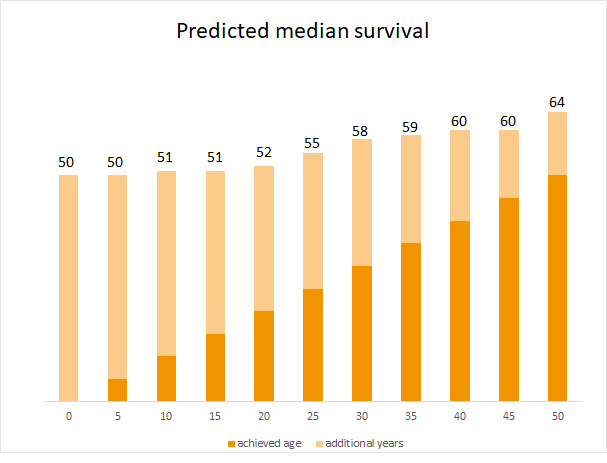
Significant progress has already been made in this area. An example is the metabolic disorder cystic fibrosis. Here, the predicted average survial of a newborn has increased from one year to 50 years by using so-called CFTR modulators.14
Current status
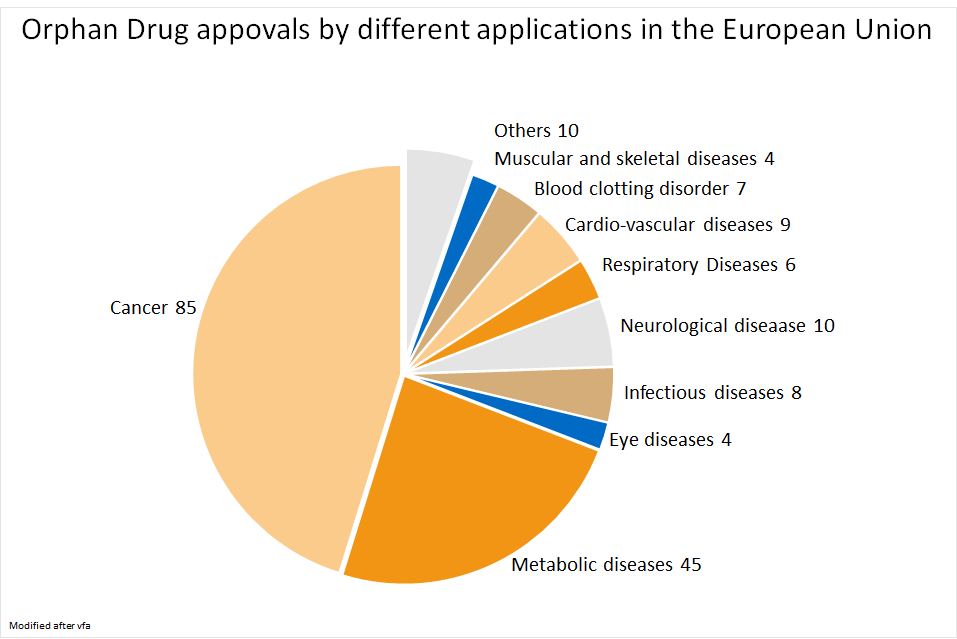
There’re 158 drugs for 131 diseases authorized in the European Union (as of 2019).3,4 Cancer treatment accounts for the largest share, followed by drugs for metabolic disorders.4
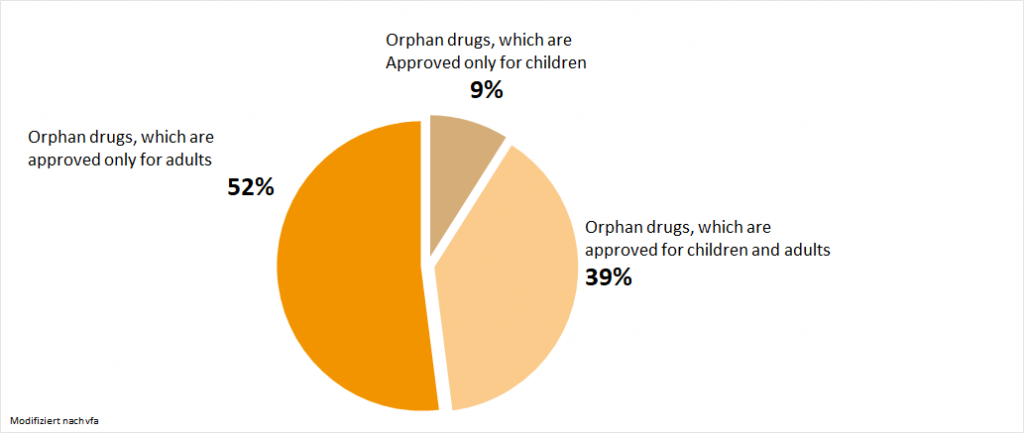
Nearly half of all drugs are also authorized for children:1
There’re 23 drugs approved for rare diseases affecting a maximum of 1,000 patients in Germany.3 A total of 93 orphan drugs were prescribed in 2017,4 which accounts for approximately 3.7% of total health insurance expenditures.1,3,4
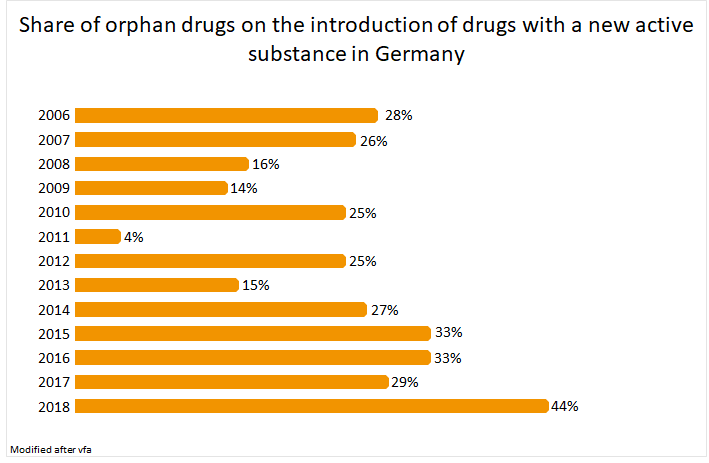
The share of orphan drugs is approximately 33% of all new drugs approved in Germany between 2014 and 2017.1,4 This number is increasing dramatically in the past year due to the increasig focus on rare diseases.1,4

So a lot has happened in this field in the past years. An important point was the decryption of the human genome in 2003 as part of the human genome project.4 Currently there’re 1,900 active substances against rare diseases in clinical developent.3
The fact that there’s still a great need for research on rare diseases will be shown by the next blog.
Contact Person:
Kristina Schraml (kristina.schraml@biovariance.com)




